Worth their salt: New battery anodes use salt for energy, stability

Rock salt can give a car a little extra traction on a snowy day, but it turns out that it might also be of benefit to fast charging batteries in electric cars as well.
Researchers at the U.S. Department of Energy’s (DOE) Argonne National Laboratory and the University of California San Diego have discovered that a material that looks geometrically similar to rock salt could be an interesting candidate for lithium battery anodes that would be used in fast charging applications.
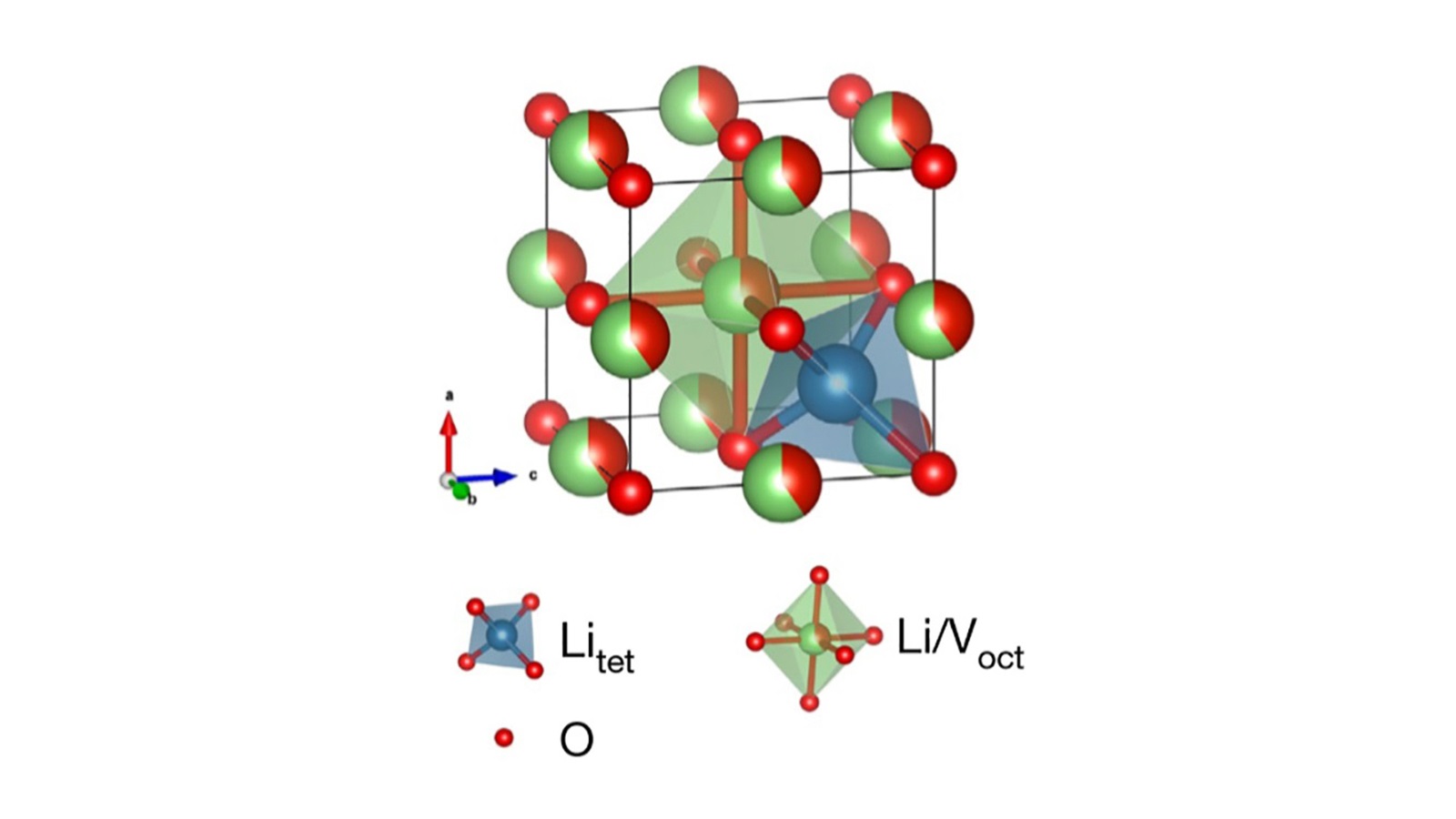
Known as a lithium rocksalt — one word — this material (Li3V2O5) contains lithium, vanadium and oxygen atoms arranged in the same way as table salt, but with more disorder in its crystal structure.
The lithium rocksalt anode offers a Goldilocks-like solution to a problem that has faced lithium battery designers. Some anodes, like graphite, are too unstable during fast charging, while others, like lithium titanate, cannot store enough energy to be fully effective.
“The capacity and energy will be a little bit lower than graphite, but it’s faster, safer and has a longer life. It has a more negative chemical potential, and therefore, much improved energy density over current commercialized fast charging lithium-titanate anodes,” said Haodong Liu, a postdoctoral associate at UC San Diego and first author of the paper.
To discover the structure and dynamics of the lithium rocksalt, the UC San Diego team collaborated with researchers at DOE’s Oak Ridge National Laboratory (ORNL), who used neutron diffraction to determine the atomic structure of the rocksalt material. Researchers at UC Irvine and DOE’s Brookhaven National Laboratory led by Professor Huolin Xin performed high resolution microscopic studies to resolve the structural changes after lithium insertion.
Once completed, the Argonne team, led by chemist Jun Lu, as well as scientists from DOE’s Lawrence Berkeley National Laboratory, conducted X-ray diffraction and X-ray absorption studies to reveal the crystal structural change and charge compensation mechanisms of the material during charging and discharging. This study used national lab facilities including the beamline VULCAN (Spallation Neutron Source at ORNL), beamline 9-BM (Advanced Photon Source (APS) at Argonne), and beamline 5.3.1 (Advanced Light Source at Berkeley Lab). All three facilities are DOE Office of Science User Facilities.
“For a battery to be able to both quickly charge and hold a great deal of energy would represent a tremendous accomplishment and researchers are constantly trying to push the frontier in these areas,” Lu said. “This new material gives us the ability to explore potential new breakthroughs or applications without as many steep trade-offs as previous technologies.”
“The capability to carry out in situ experiments at 9-BM made a significant impact on this project,” added Argonne physicist Tianpin Wu. “By taking X-ray absorption spectroscopy data, the researchers were able to catch all changes inside the battery during charging and discharging cycles, and the high brilliance of the X-rays at the APS ensured outstanding data quality.”
A paper based on the study, “A disordered rock salt anode for fast-charging lithium-ion batteries,” appeared in Nature on Sept. 2. Authors include Haodong Liu, Zhuoying Zhu, Qizhang Yan, Sicen Yu, Yiming Chen, Yejing Li, Xing Xing, Yoonjung Choi, Shyue Ping Ong and Ping Liu from UC San Diego; Xin He, Jun Feng and Robert Kostecki from Berkeley Lab; Yan Chen and Ke An from ORNL; Rui Zhang and Huolin Xin from the University of California; Lu Ma and Ruoqian Lin from Brookhaven Lab; Tongchao Liu, Matthew Li, Khalil Amine, Tianpin Wu and Jun Lu from Argonne; Lucy Gao from Del Norte High School and Helen Sung-Yun Cho from Canyon Crest Academy.
About the Advanced Photon Source
The U. S. Department of Energy Office of Science’s Advanced Photon Source (APS) at Argonne National Laboratory is one of the world’s most productive X-ray light source facilities. The APS provides high-brightness X-ray beams to a diverse community of researchers in materials science, chemistry, condensed matter physics, the life and environmental sciences, and applied research. These X-rays are ideally suited for explorations of materials and biological structures; elemental distribution; chemical, magnetic, electronic states; and a wide range of technologically important engineering systems from batteries to fuel injector sprays, all of which are the foundations of our nation’s economic, technological, and physical well-being. Each year, more than 5,000 researchers use the APS to produce over 2,000 publications detailing impactful discoveries, and solve more vital biological protein structures than users of any other X-ray light source research facility. APS scientists and engineers innovate technology that is at the heart of advancing accelerator and light-source operations. This includes the insertion devices that produce extreme-brightness X-rays prized by researchers, lenses that focus the X-rays down to a few nanometers, instrumentation that maximizes the way the X-rays interact with samples being studied, and software that gathers and manages the massive quantity of data resulting from discovery research at the APS.
This research used resources of the Advanced Photon Source, a U.S. DOE Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under Contract No. DE-AC02-06CH11357.
Argonne National Laboratory seeks solutions to pressing national problems in science and technology by conducting leading-edge basic and applied research in virtually every scientific discipline. Argonne is managed by UChicago Argonne, LLC for the U.S. Department of Energy’s Office of Science.
The U.S. Department of Energy’s Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information, visit https://energy.gov/science.