An innovative twist on quantum bits: Tubular nanomaterial of carbon makes ideal home for spinning quantum bits
Scientists are vigorously competing to transform the counterintuitive discoveries about the quantum realm from a century past into technologies of the future. The building block in these technologies is the quantum bit, or qubit. Several different kinds are under development, including ones that use defects within the symmetrical structures of diamond and silicon. They may one day transform computing, accelerate drug discovery, generate unhackable networks and more.
Working with researchers from several universities, scientists at the U.S. Department of Energy’s (DOE) Argonne National Laboratory have discovered a method for introducing spinning electrons as qubits in a host nanomaterial. Their test results revealed record long coherence times — the key property for any practical qubit because it defines the number of quantum operations that can be performed in the lifetime of the qubit.
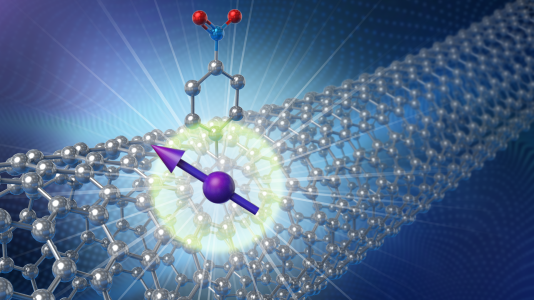
Electrons have a property analogous to the spin of a top, with a key difference. When tops spin in place, they can rotate to the right or left. Electrons can behave as though they were rotating in both directions at the same time. This is a quantum feature called superposition. Being in two states at the same time makes electrons good candidates for spin qubits. Spin qubits need a suitable material to house, control and detect them, as well as read out information in them. With that in mind, the team chose to investigate a nanomaterial that is made from carbon atoms only, has a hollow tubular shape and has thickness of only about one nanometer, or a billionth of a meter, roughly 100,000 times thinner than the width of a human hair.
“Much to our gratification, our chemical modification method creates an incredibly stable spin qubit in a carbon nanotube.” — Jia-Shiang Chen, chemist at Center for Nanoscale Materials at Argonne and Center for Molecular Quantum Transduction at Northwestern University
“These carbon nanotubes are typically a few micrometers long,” said Xuedan Ma. “They are mostly free of fluctuating nuclear spins that would interfere with the spin of the electron and reduce its coherence time.”
Ma is a scientist in Argonne’s Center for Nanoscale Materials (CNM), a DOE Office of Science user facility. She also holds appointments at the Pritzker School of Molecular Engineering at the University of Chicago and Northwestern-Argonne Institute of Science and Engineering at Northwestern University.
The problem the team faced is that carbon nanotubes by themselves cannot maintain a spinning electron at one site. It moves about the nanotube. Past researchers have inserted electrodes nanometers apart to confine a spinning electron between them. But this arrangement is bulky, expensive and challenging to scale up.
The current team devised a way to eliminate the need for electrodes or other nanoscale devices for confining the electron. Instead, they chemically alter the atomic structure in a carbon nanotube in a way that traps a spinning electron to one location.
“Much to our gratification, our chemical modification method creates an incredibly stable spin qubit in a carbon nanotube,” said chemist Jia-Shiang Chen. Chen is a member of both CNM and a postdoctoral scholar in the Center for Molecular Quantum Transduction at Northwestern University.
The team’s test results revealed record long coherence times compared to those of systems made by other means — 10 microseconds.
Given their small size, the team’s spin qubit platform can be more easily integrated into quantum devices and permits many possible ways to read out the quantum information. Also, the carbon tubes are very flexible and their vibrations can be used to store information from the qubit.
“It is a long way from our spin qubit in a carbon nanotube to practical technologies, but this is a large early step in that direction,” Ma said.
The team’s findings were reported in Nature Communications. In addition to Ma and Chen, authors include Matthew D. Krzyaniak and Michael R. Wasielewski (Northwestern University), Kasidet Jing Trerayapiwat and Sahar Sharifzadeh (Boston University), Lei Sun (Westlake University) and Tijana Rajh (Arizona State University).
The work was supported by the DOE Office of Basic Energy Sciences and National Science Foundation.
About Argonne’s Center for Nanoscale Materials
The Center for Nanoscale Materials is one of the five DOE Nanoscale Science Research Centers, premier national user facilities for interdisciplinary research at the nanoscale supported by the DOE Office of Science. Together the NSRCs comprise a suite of complementary facilities that provide researchers with state-of-the-art capabilities to fabricate, process, characterize and model nanoscale materials, and constitute the largest infrastructure investment of the National Nanotechnology Initiative. The NSRCs are located at DOE’s Argonne, Brookhaven, Lawrence Berkeley, Oak Ridge, Sandia and Los Alamos National Laboratories. For more information about the DOE NSRCs, please visit https://science.osti.gov/User-Facilities/User-Facilities-at-a-Glance.
Argonne National Laboratory seeks solutions to pressing national problems in science and technology by conducting leading-edge basic and applied research in virtually every scientific discipline. Argonne is managed by UChicago Argonne, LLC for the U.S. Department of Energy’s Office of Science.
The U.S. Department of Energy’s Office of Science is the single largest supporter of basic research in the physical sciences in the United States and is working to address some of the most pressing challenges of our time. For more information, visit https://energy.gov/science.